CUSTOMER SUCCESS STORY
Washington University: Dual-Laser Multiphoton Microscope for Immunology and Other Research
The Challenge
Dr. Mark Miller, Associate Professor and Director of the In Vivo Imaging Core at Washington University School of Medicine in St. Louis, has a long history of building and using multiphoton microscopes, particularly for use in intravital studies of immunity. This includes landmark investigations into leukocyte trafficking and antigen presentation and making novel use of computational biology. For example, his group first revealed that the swarming of T-cells relative to APC (antigen-presenting cell) targets was not dominated by chemokine gradients but rather commenced with random movements (Miller et al., Science 2002, Miller et al., PNAS, 2004, Miller et al. JEM, 2004).
Since 2012 he has also acted as the Director of the In Vivo Imaging Core where he set out to build a universal multiphoton microscope with open access for interdisciplinary researchers to perform single-cell imaging in studying infectious diseases in vivo. A key goal was to include two independently tunable wavelengths (Zinselmeyer et al., Methods Enzymology, 2009) to enable flexible experiments with multiple fluorophores. Other prerequisites were laser ease of use to support a wide user base, and high reliability to enable efficient multi-user scheduling.
The Solution
Dr. Miller eventually chose to modify a commercially available scope and equip it with two Chameleon Vision II lasers, in part because of extensive positive experience with previous Chameleon Ultra lasers in his immunology lab. He adds that he chose the Vision II models as these provided the longest wavelength direct tuning range of any titanium:sapphire laser. “This gives our collaborators a full range of excitation options including 800 nm for autofluorescence, 900 nm for green- (and cyan-) fluorescent proteins, and 1040 nm for use with red fluorescent proteins of the mfruit series. The long wavelength range is also useful for minimizing autofluorescence and photodamage in live tissue experiments needing long acquisition times.”
Dr. Miller notes that the innovative service/replacement contract was another key factor in the laser choice. “We’d had a couple of issues with the early-generation lasers. But Coherent immediately brought us back to 100% specified performance, with the only downtime literally being the time to ship a replacement laser to my lab.” While he has had no problems at all with the Vision II lasers, he explains that nonetheless the contract provides invaluable “peace of mind” and allows the non-stop running of the busy imaging core.
The Result
Dr. Miller states that the dual-laser microscope has been an unqualified success in terms of performance, reliability, and operational simplicity, where new users typically can operate the system after just a few hours of hands-on training This instrument has produced data for dozens of publications that cover a diverse range of topics. Dr. Miller has posted an impressive presentation on YouTube that includes a host of images and videos from his long history of using the intravital multiphoton imaging method.
References:
Two-photon imaging of lymphocyte motility and antigen response in intact lymph node.
Miller MJ, Wei SH, Parker I, Cahalan MD. Science. 2002 Jun 7;296(5574):1869-73. doi: 10.1126/science.1070051. Epub 2002 May 16.
T cell repertoire scanning is promoted by dynamic dendritic cell behavior and random T cell motility in the lymph node. Miller MJ, Hejazi AS, Wei SH, Cahalan MD, Parker I. Proc Natl Acad Sci U S A. 2004 Jan 27;101(4):998-1003. doi: 10.1073/pnas.0306407101. Epub 2004 Jan 13.
Imaging the single cell dynamics of CD4+ T cell activation by dendritic cells in lymph nodes.
Miller MJ, Safrina O, Parker I, Cahalan MD. J Exp Med. 2004 Oct 4;200(7):847-56. doi: 10.1084/jem.20041236.
Chapter 16. Two-photon microscopy and multidimensional analysis of cell dynamics.
Zinselmeyer BH, Dempster J, Wokosin DL, Cannon JJ, Pless R, Parker I, Miller MJ.
Methods Enzymol. 2009;461:349-78. doi: 10.1016/S0076-6879(09)05416-0.
“The innovative service contract means that the only downtime is literally the time to ship a replacement laser to my lab."
— Mark Miller, Associate Professor, Department of Medicine, Washington University School of Medicine in St. Louis


Figure 1. The WashU IVIC custom-built two-photon system
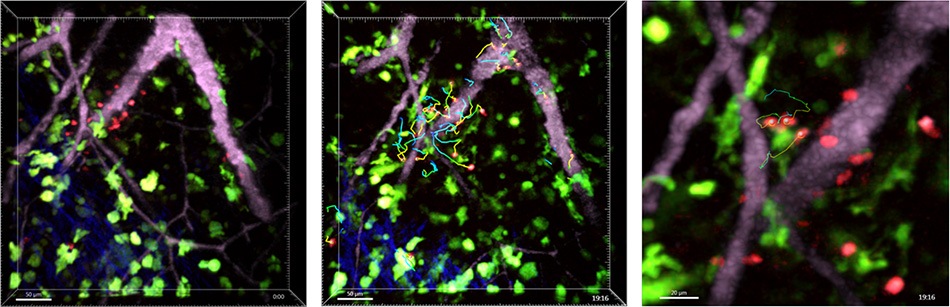
Figure 2. In vivo two-photon imaging of “T cell antigen recognition” reveals the key role played by random T cell motility in initiating adaptive immune responses. T cells (red) in the cervical lymph node migrate away from high endothelial venules(pink) and make random contacts with dendritic cells (green, zoomed view).