Optogenetics – Controlling Cells and Animals with Light
Optogenetics lets researchers activate, de-activate, and manipulate nerve cells just using light, usually from lasers.
February 8, 2022 by Coherent
Over three hundred years ago, Luigi Galvani discovered that applying electricity to a frog’s leg caused it to twitch. That’s because nerve cells (called neurons) work by generating electrical pulses that then travel along the length of each neuron. The electrical pulse is called the “action potential.”
Scientists have long used tiny electrodes to apply electrical pulses to neurons to force them to fire on command. They used this to investigate how some of the neurons are connected together, and even to map out which parts of the brain control which parts of the body.
But, the brain does way more than control our physical movement. It thinks and remembers, and processes all the incoming signals from our sensory organs (eyes, ears, etc.). Up to about 25 years ago, we didn’t know much about how the brain performed even the most ordinary of these tasks – like recalling a name or recognizing a friend’s face.
That’s changing very fast now. Scientists are using all sorts of tools and techniques to study the brains of animals. The mouse, in particular, is a commonly used test subject, because it’s a mammal with a similar basic brain structure to human beings.
Optogenetics lights up neuroscience
The latest method in neuroscientists’ toolbox is called optogenetics. It causes nerves to fire using light instead of an electrode. To accomplish this, scientists use a particular kind of protein molecule called an opsin. Specifically, these are proteins that respond when light hits them.
In 2005, neuroscientists Karl Deisseroth, Ed Boyden, and their colleagues showed that you could genetically engineer animals, including mice, to develop nerve cells that incorporate opsins and thus become sensitive to light stimuli. What’s more, they precisely manipulated the genetic engineering so that they could choose exactly what types of nerve cells "express” which kinds of opsins.
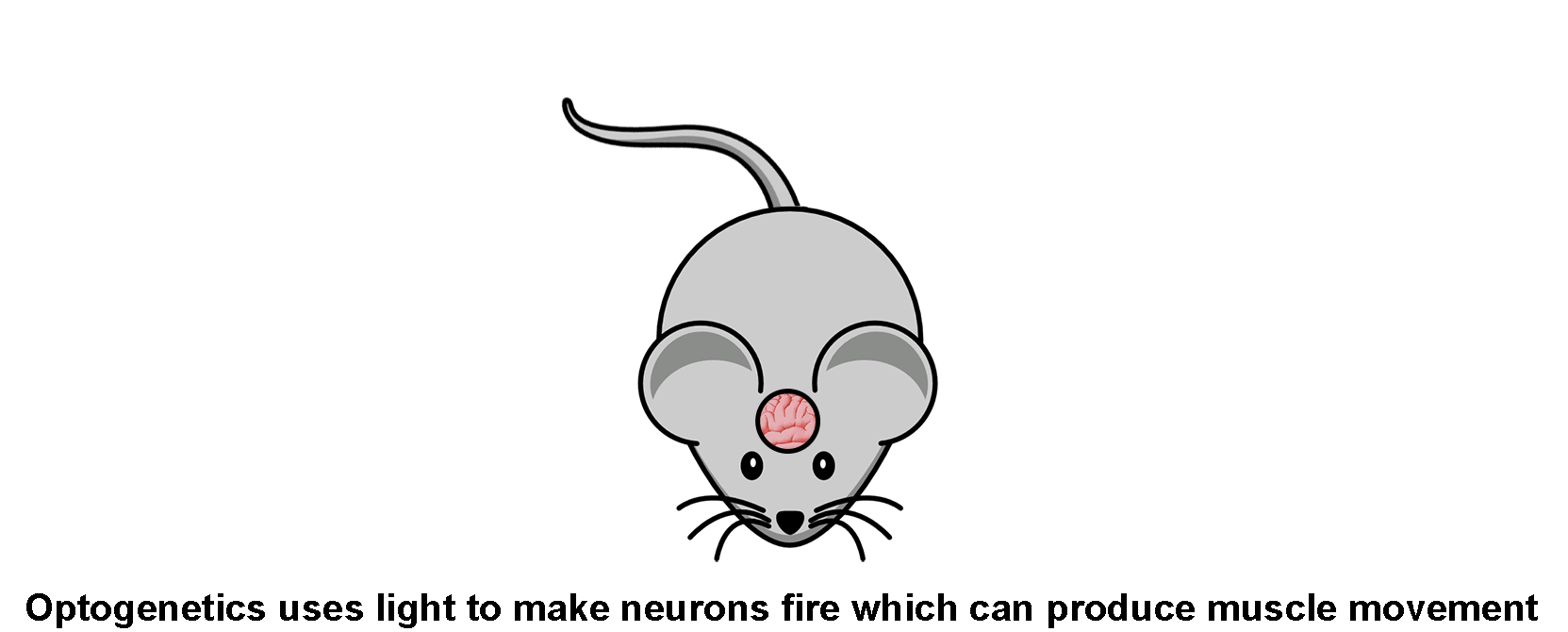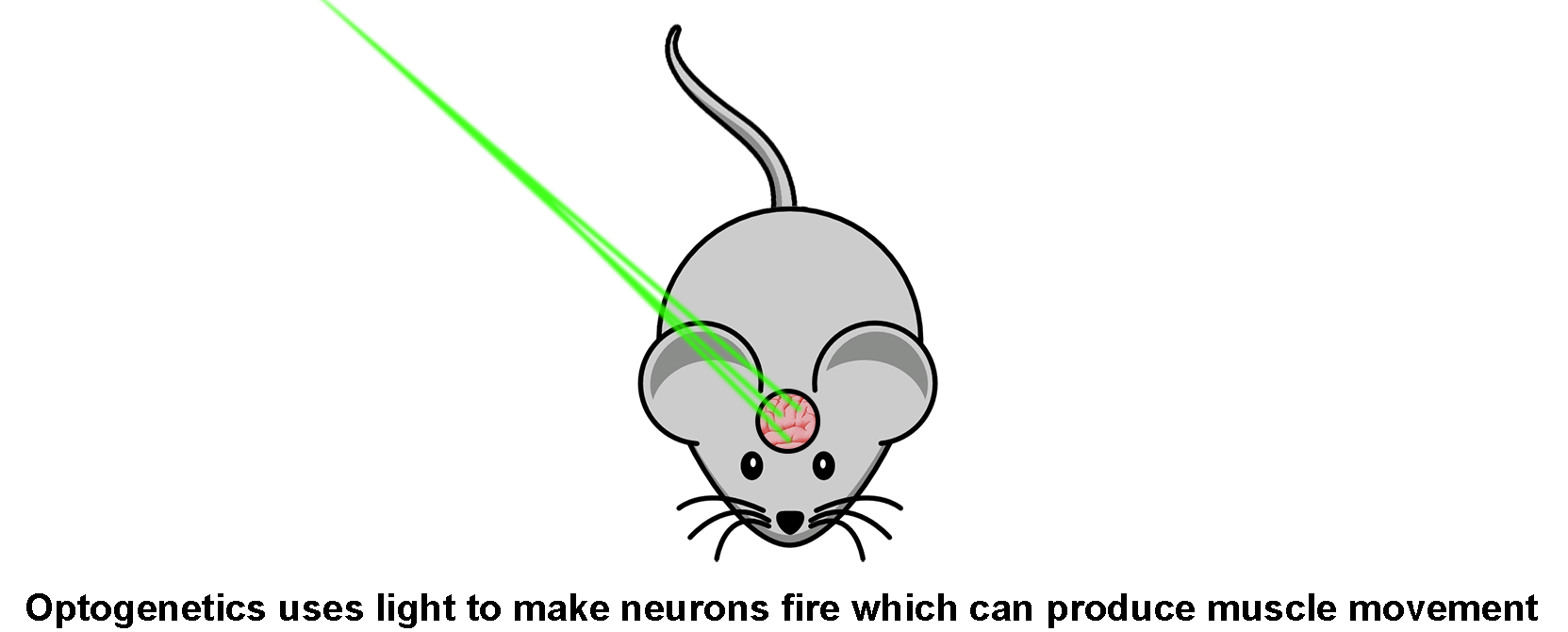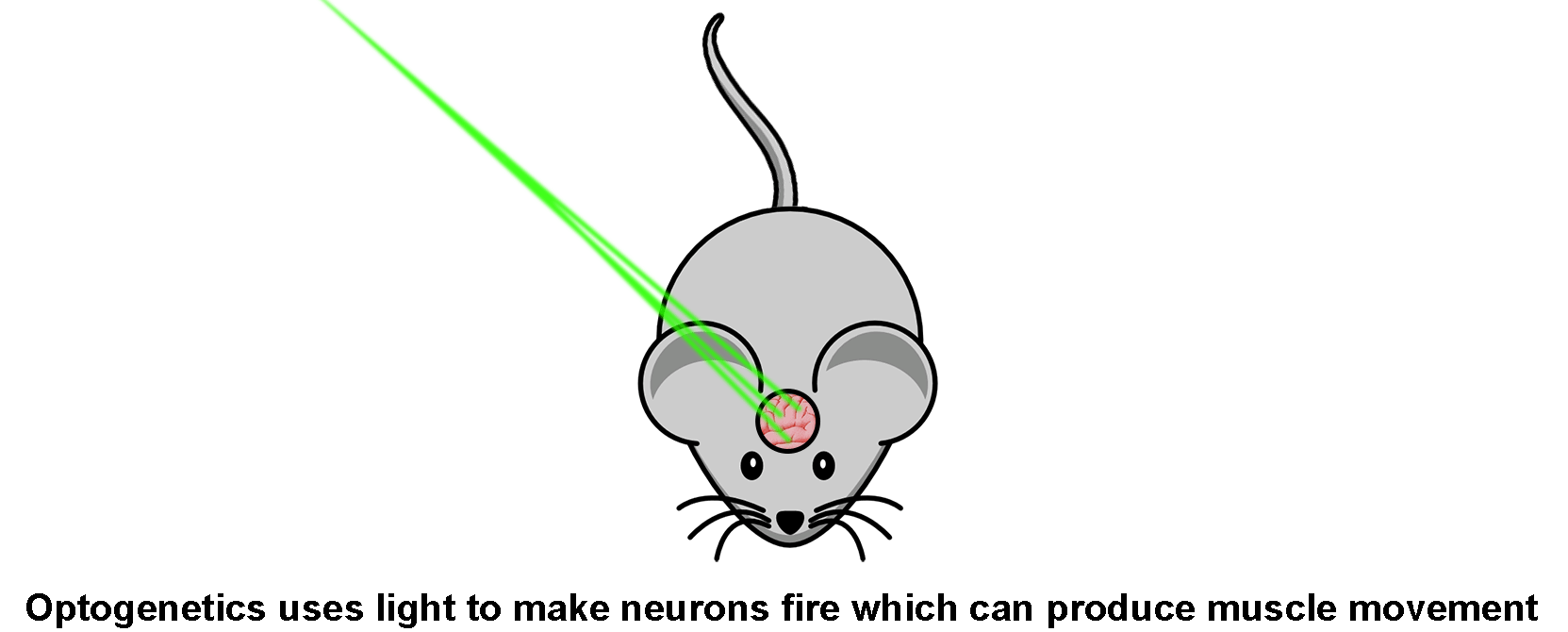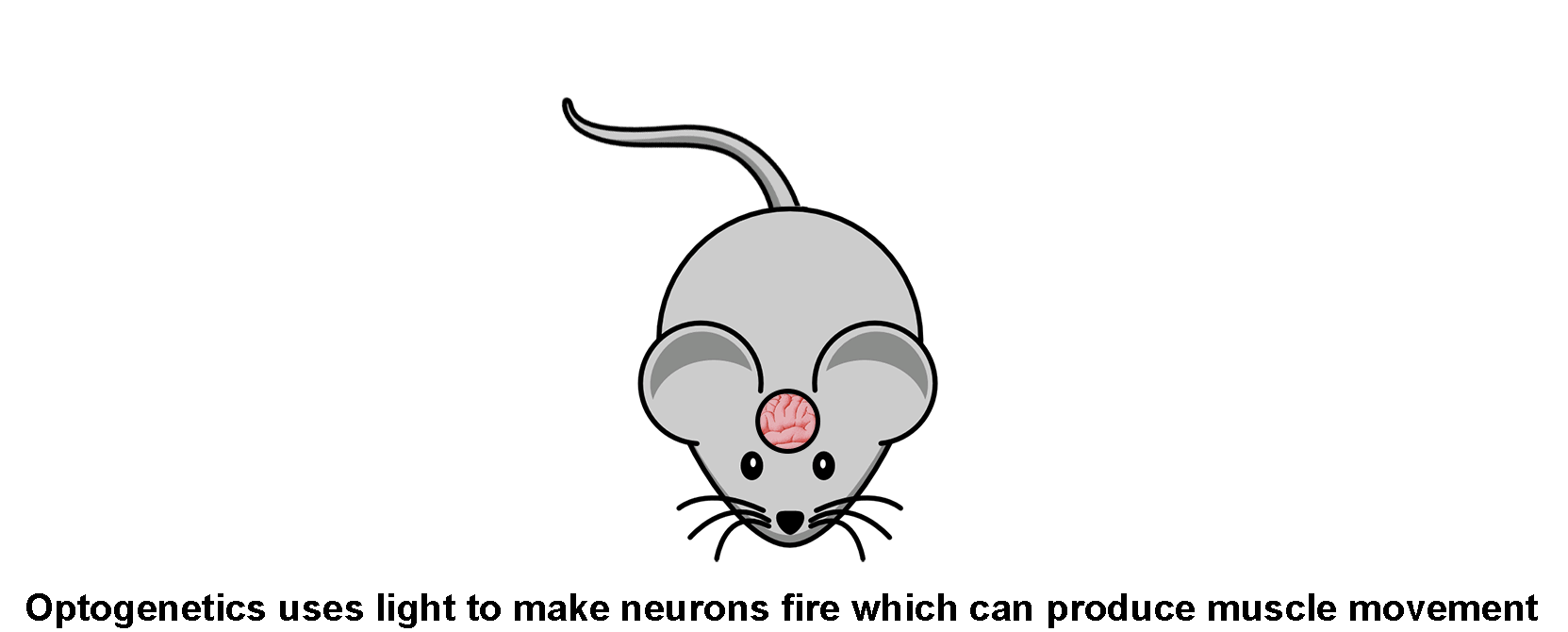
And just like that, you could now fire nerve cells in a live mouse simply by shining light of a certain color on them, rather than by physically connecting electrodes to the nerves. Alternatively, you could use different opsins to prevent a nerve from firing by shining another color of light on them. That’s what optogenetics is.
Lasers provide a light touch for optogenetics
The big advantage of optogenetics is that light is a non-contact and selective tool that is far less intrusive than a physical electrode. And light can be quickly moved from place to place on the subject animal without harming it. Plus, if it’s laser light, it can be focused down to a small spot so that only a very specific part of the brain is stimulated (or de-activated).
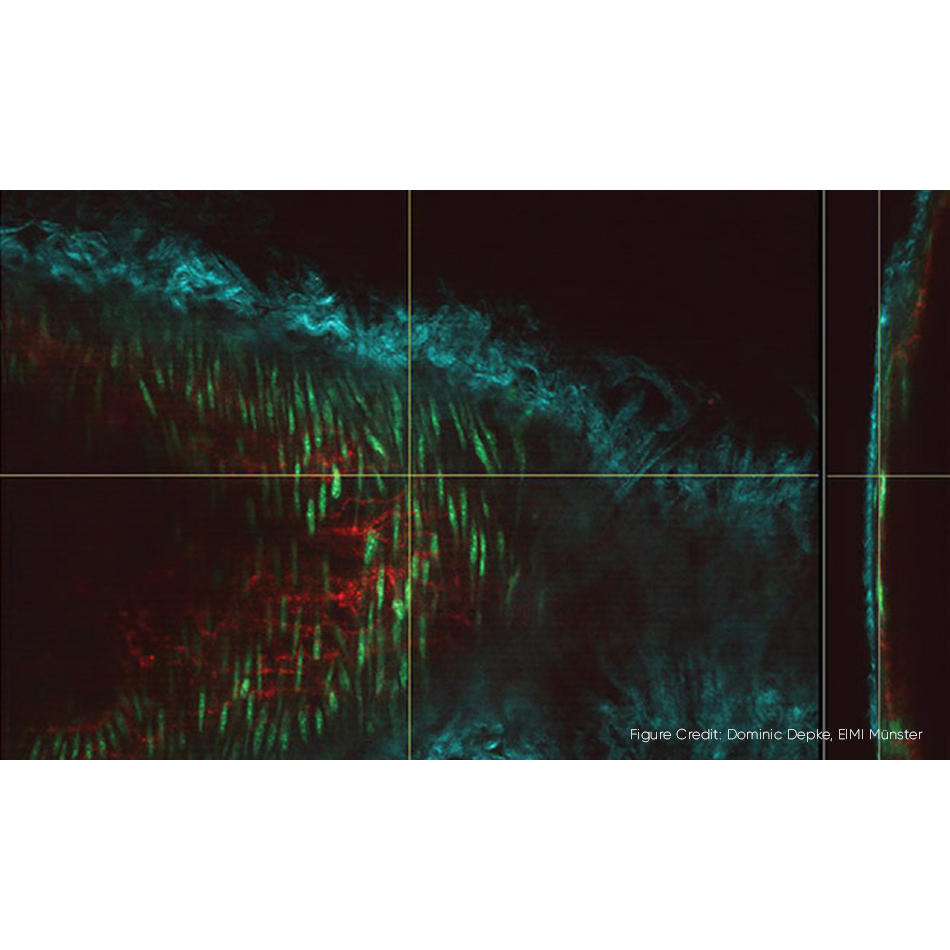
The most advanced optogenetic experiments use ultrafast lasers, like the Coherent Monaco. The advantage of ultrafast lasers is that they can be used to selectively excite individual neurons in a mouse’s brain by utilizing an effect called “multiphoton excitation.” Plus, the infrared light used for this technique penetrates efficiently into the brain cortex.
Increasingly, neuroscientists project a pattern of multiple laser spots controlled by computer algorithms. This enables them to stimulate hundreds of precisely targeted neurons and see exactly how they interact with each other. Following how this actually happens is done using various multiphoton excitation (MPE) microscopy techniques, based on lasers like the Coherent Axon or Chameleon series combined with Monaco.
Eventually, neuroscientists will be able to measure how much larger numbers of neurons communicate and interact with each other in real-time. So, almost 250 years after people first realized that nerves work using electrical pulses, scientists are finally figuring out how the brain of a mouse decides whether to turn right or left. Now, if we could just remember where we left our car keys...
Related Resources