ARTICLE
Laser Modulation Solutions
for Two-Photon Microscopy
Overview
Since the seminal work in two-photon laser scanning fluorescence microscopy published in 1990 (Denk, et al., 1990), the technique has benefitted from step changes in laser technology. These improvements have furthered the penetration of the technique initially from a physics laboratory, to cell biology, disease studies, and advanced neuroscience imaging.
One box, tunable Ti:Sapphire lasers started this trend around 2001. Some years later, automatic dispersion control was added to the lasers to optimize the pulse duration at the sample plane of the microscope. As probes excitable at wavelengths longer than the upper limit of Ti:Sapphire lasers became more mature and efficient, after 2010 laser companies turned to Optical Parametric Oscillators to address this need for an augmented color palette, deeper imaging, and less photodamage.
In this article, we discuss the next phase in this evolution; the integration of fast power modulation in the laser system, and how this enables faster setup time, highest performance, and low cost of ownership.
Requirements for Laser Power Control in Two-Photon Microscopy.
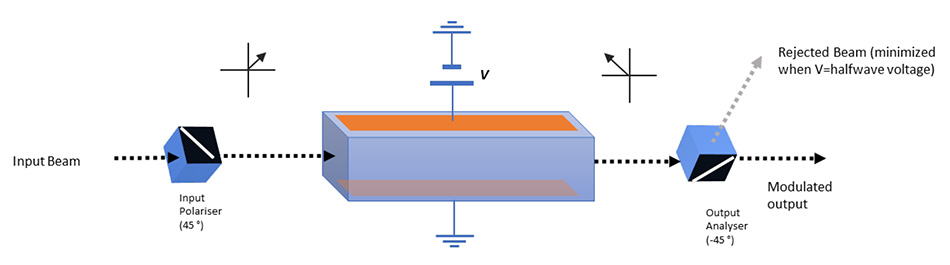
In its simplest form, continuous control of laser power can be achieved by addition of a phase retarding waveplate and polarizing analyser. By rotation of the waveplate, the transmission of the laser power through the analyser can typically be changed from 0.2% transmission to ~99%. By motorizing the waveplate this process can be automated to alter the power at the imaging plane in the microscope to equalize focused fluences at different depth frames, for example.
Most modern laser scanning two-photon microscopes, however, require a faster modulation speed. For example, for a raster laser scanning application where data acquisition should be made in a single direction only, the laser must be blanked during the “flyback operation” to avoid unwanted fluorescence excitation or photobleaching. In the case of resonant galvometric scanners, the resulting rise/fall times can be as small as a few microseconds. In this domain, one must consider optical modulation methods.
Electro-optical Modulation
An Electro-Optic Modulator (EOM) modulates laser power by applying a phase retardation to the beam by use of the Pockels effect. This is where birefringence is induced in a non- centrosymmetric crystal by application of an electric field. As before, a polarizing analyzer is used to complete the modulator setup.
Pockels cells may be configured in a longitudinal excitation geometry, to accommodate larger beams with relatively short crystals. In this case the typical 1⁄2 wave voltage (i.e the voltage required for a 90-degree rotation of the polarization), is in the order of 6 kV, which is difficult to achieve at the speeds and duty cycles required for 2P microscopy. Thus, most configurations for imaging adopt the transverse electric field geometry, using longer crystals, which significantly lowers the halfwave voltage. Crystals are generally deployed in 2 or more serial configurations, rotated with respect to each other, to further lower the required switching voltage and compensate for thermal loading effects.
Care must be taken to optimize pulse contrast (ratio of minimum versus maximum transmitted power) by crystal alignment and offset (bias) voltage adjustment for best image contrast.
Pockels cells are widely adopted in two-photon microscopy, especially amongst the “home builders” community as they are relatively simply deployed, especially for users requiring only modest powers at popular two-photon wavelengths.
For example, cells based on potassium dideuterium phosphate (KD*P) offer excellent transmission, speed, and contrast characteristics for 2P applications up to approximately 1100nm, and modest laser powers. Additionally, KD*P has low group velocity dispersion characteristics, resulting in minimal group delay dispersion (GDD). To this end KD*P Pockels cells are a popular choice when using ultrafast lasers without dispersion precompensation and limited tuning, such as Ti:Sapphire lasers.
Acousto-optic Modulation
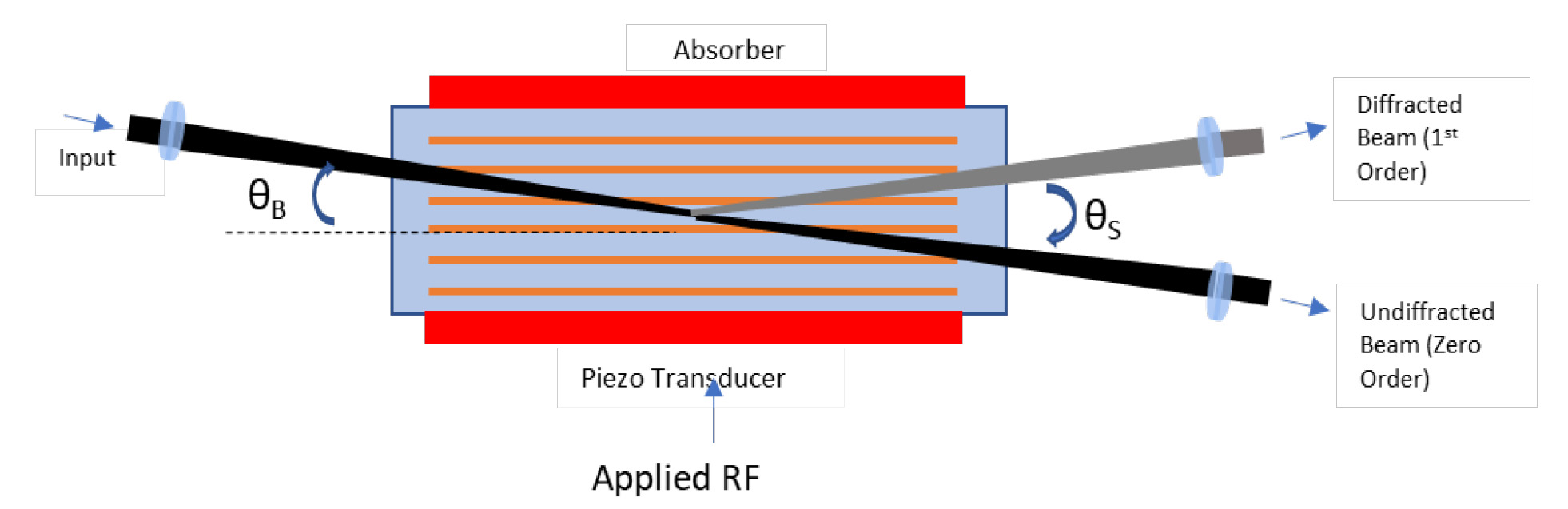
An Acousto-Optic Modulator (AOM) comprises a transparent crystal or glass onto which a piezoelectric transducer is attached. A radiofrequency (RF) wave applied to the transducer induces an acoustic wave that strains the crystal resulting in a refractive index grating. Light travelling through the cell then undergoes Bragg diffraction.
The rise/fall time achievable is proportional to the time the acoustic wave takes to cross the laser beam, thus is optimized by reducing the beam’s width in the crystal.
Discrimination and therefore contrast ratio is defined by both the separation angle (θS) between the zero and first diffraction orders and the distance to the working plane of interest.
The most common AOM material used in two-photon microscopy is tellurium dioxide, (TeO2). This material demonstrates excellent diffraction efficiency and high-power handling over a wide wavelength range. Maximum transmission efficiencies are achieved with modest RF powers in the order of 30 dBm.
TeO2 AOMs are usually configured in the Bragg interaction regime which offers best diffraction efficiency into the first order, with higher orders destructively annihilated. Note that to achieve high efficiency with minimal RF power levels, crystal lengths of >1 cm are needed, resulting in non-negligible group delay dispersion (GDD). Considering also the dispersion of other downstream optics and especially the objective lens, AOM-based microscope systems benefit by being partnered with lasers equipped with dispersion precompensation, in order to maintain the shortest pulses at the sample plane.
Deployment of AOMs for tunable lasers requires both careful optical and control electronics design. Since the separation angle (θs) is dependent on both the RF drive frequency (ie grating period) and the laser wavelength, the RF drive frequency must be carefully calibrated to ensure minimal pointing change when tuning the laser wavelength. Additionally, the maximum diffraction efficiency is achieved at different RF powers for different wavelengths. The more onerous integration effort resulting from the need to carefully control the RF frequency and power, and manage a relatively large GVD in a tunable imaging system has hitherto limited AOM usage in many home-builder and custom settings, despite the excellent performance characteristics.
Modulation in Widely tunable lasers
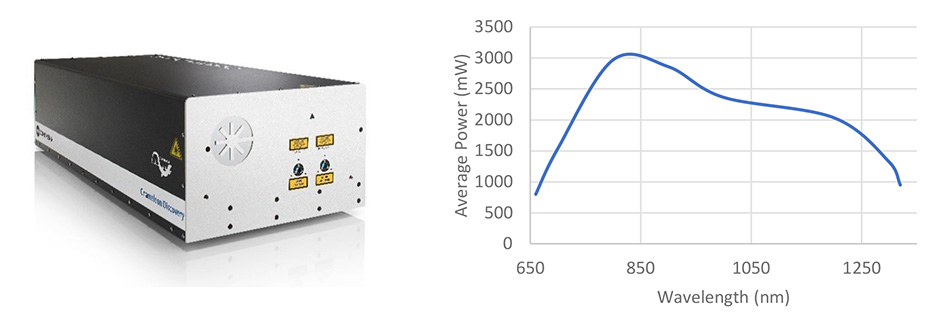
The advent of one-box widely tunable lasers, in the order of 680-1300 nm and with powers in excess of 2 W, requires a new regime of performance and integration effort for laser modulation.
The typically used KD*P Pockels cells display thermal blooming effects at high power, which is deleterious to beam pointing, beam waist integrity, and lifetime. Longer wavelengths further present higher drive voltage and contrast challenges. Lithium Tantalate is a viable EOM material for wider tuning, however the group delay dispersion of commercial units is higher than the correctable range of dispersion-compensated lasers, thus resulting in longer pulses and reduced peak power, detrimental to efficient imaging.
As previously discussed, despite having potential cost and performance benefits, AOM-based solutions require a high degree of optical design and electronics control expertise to deploy, often not readily available in many bioimaging facilities. That said, AOM solutions are commercially available as an integrated solution from some microscope vendors.
In 2017, Coherent recognized that both users and the microscope industry would benefit from a turn-key solution integrating the AOM modulation with the laser sources. Building on the expertise gleaned from integrated AOM solutions in industrial ultrafast machining lasers, Coherent developed Total Power Control (TPC) – as a fully integrated option for the Chameleon Discovery Laser.
Total Power Control, available on Chameleon Discovery NX, provides high contrast (>1000:1) and high speed (<1 μs rise time) modulation across a full octave tuning range of 660 nm to 1320 nm in a hands-free automated package.
All the taxing requirements for RF frequency and power calibration and adjustment are programmed internally to the laser, so all the user or microscope integrator needs to provide is the set wavelength and power level required.
Since AOMs are very cost-effective, the fixed wavelength 1040 nm output of Chameleon Discovery NX TPC is also equipped with its own, dedicated AOM and driver. Power can be conveniently controlled by either serial/USB command or by fast analog control input.
Future trends
As the scope of two-photon imaging techniques drives further into OEM and preclinical applications, the demand for single wavelength, cost-effective femtosecond sources is growing. The Axon series of compact ultrafast sources addresses perfectly these requirements.
From product concept stage, TPC capability was integrated into the Axon design to simplify deployment into new microscope designs and applications. This brings ultimate integration convenience for applications where the two-photon microscope system is part of a movable diagnostic, clinical or high-throughput screening device rather than a pure research instrument.
In cutting-edge neuroscience research, high-power lasers are playing a key role in all-optical in- vivo imaging techniques, using optogenetic stimulation (Yuste, 2012). Multiple tens of watts of laser power are split with spatial light modulators (SLMs) into individual beamlets able to individually address tens or hundreds of neurons. This method of optical control requires short and tailorable burst of pulses. High-power fiber lasers like the Coherent Monaco provide the flexibility demanded by these applications thanks to the all-fiber design format. The resulting high average power, high energy laser requirements, and the need to switch the stimulation beam on a sub-millisecond timescale presents a specific challenge for incumbent Pockels cell technology. To this end, AOM technology has been fully integrated into Monaco, for exquisite pulse control, simplified microscope design, and increased imaging system reliability.



Conclusions
In this technical note, we have discussed the two leading approaches to modulate the laser output power of femtosecond lasers used in two-photon microscopy – Electro-Optic and Acousto- Optic modulation. Most “home-builders” have so far chosen EOMs because of the relative simplicity of deploying this high-voltage powered device in the optical path. Various microscope vendors offer either EOMs or AOMs partially integrated in their laser delivery train, with their software architecture controlling both microscope and laser. Using its manufacturing experience with high-power fiber lasers designed for 24/7 manufacturing environments, Coherent recognized that the advantages of the AOM approach in term of size, cost, speed, and overall performance would also fulfill two-photon imaging applications. By integrating the sophisticated control of the AOM within the laser software and hardware architecture of Discovery NX, Axon, and Monaco, two-photon users – both home builders and scope companies - benefit from a greatly simplified and easier to control optical setup, for applications ranging from advanced neuroscience to medical diagnostics.
Works Cited
W. Denk, J.H. Strickler, and W.W. Webb Two-photon laser scanning fluorescence microscopy. Science - 1990. - pp. 73-76.
A.M Packer, D.S. Peterka, J.J. Hirtz, R. Prakash, K. Deisseroth and R. Yuste Two-photon optogenetics of dendritic spines and neural circuits. Nat Methods, 9 (12), 1205-1205, 2012.